|
It helps in the detection of various other metal ions apart from nickel. The formula of dimethylglyoxime is C 4 H 8 N 2 O 2 , and it is also known as 2,3-Butanedione dioxide.DMG is used as a reagent in analytical chemistry, and its multiple derivatives have been synthesised. By the end of this discussion, you will also have an idea about its structure, properties along with uses.Dimethylglyoxime or DMG is a white-coloured powder like chemical which is substantially less soluble in water but soluble in sodium hydroxide solution or methanol.
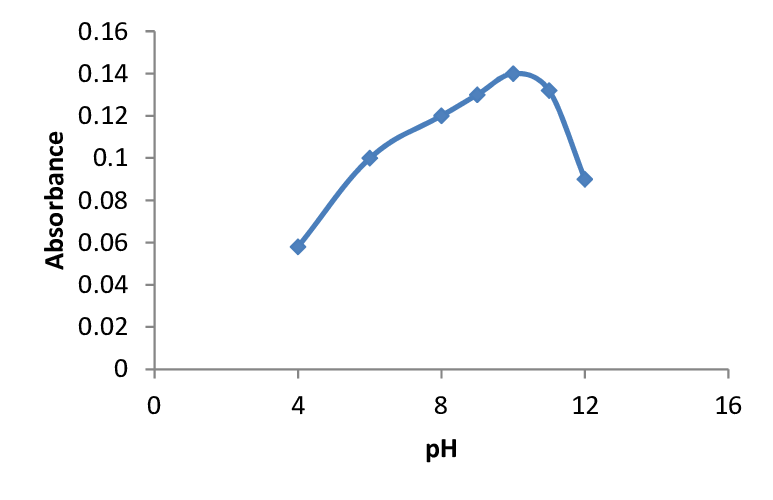
Dmg Palladium Nickel Skin Sensitivity KnownHere, dimethyglyoxime is the chelating agent.Note: Chelating agents are such organic compounds that form coordinate covalent bonds with metal ion through cationic side chains.The precipitation of nickel dimethylglyoxime is obtained with the addition of alcoholic solution of dimethylglyoxime. In this instance, the organic functional groups that precipitate with the metal ion is facilitated by chelating agents. The broad principles in the gravimetric estimation are – (1) precipitation of nickel from solution by addition of dimethylglyoxime, (2) filtering out the precipitate, and (3) calculation of the mass of nickel from the precipitate mass. Insoluble in water.Stable (incompatible with strong oxidising agents)2,3-butanedionedioxime 2,3-diisonitrosobutane diacetyldioximeDimethylglyoxime and nickel cation reacts to form red precipitation of nickel dimethylglyoxime which is insoluble in nature.Ni 2+ + 2C 4 H 8 N 2 O 2 → Ni(C 4 H 7 N 2 O 2 ) 2 + 2H +It is a common reagent in the gravimetric estimation of nickel. Physical Properties of DimethylglyoximeSoluble in alcohol, ether, pyridine, acetone. The process is highly useful in the identification of a particular kind of dermatitis or skin sensitivity known as Nickel itch.1.While a little excess of the reagent will not have any substantial impact, a significant excess can cause the reagent to precipitate. In the case of lower pH, the formation of nickel (II) ion takes place, causing the dissolution of Ni(DMG) 2. The ammonia solution helps in preventing the pH from falling below 5. Here, the electron pairs of four nitrogen atoms are donated, as opposed to the electrons of the oxygen atoms.On the other hand, precipitation from ammoniacal nickel solution takes the form of a bright red voluminous compound.DMG helps in the detection of nickel to dermatitis or similar skin ailment. In case of palladium, the precipitation takes the form of a yellow compound after diluting it with hydrochloric acid. Complete precipitation takes place after the solution stands overnight.Different Applications of DimethylglyoximeIt may be utilised as a precipitant for palladium and nickel. Subsequently, palladium is precipitated with dimethylglyoxime.The complex is treated with aqua regia, and the nitrate ions are eliminated from the resulting solution by concentrated hydrochloric acid. Palladium chloride is purified with the removal of any trace of platinum in the form of ammonium chloroplatinate. The amount of reagent to be added is directly proportional to the quantity of ions present.Ferrous sulphate and ammonium hydroxide react in the presence of dimethylglyoxime to form a compound of iron and ammonium sulphate, and at the same time releasing water.FeSO 4 + 2NH 4 OH + 2C 4 H 8 N 2 O 2 → Fe(C 4 H 7 N 2 O 2 ) 2 + (NH 4 ) 2 SO 4 + 2H 2 ODimethylglyoxime helps in the precipitation of palladium which is utilised to prepare a standard palladium solution out of palladium chloride. 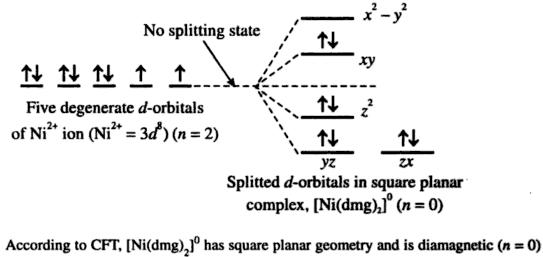
In the instance of a fire, highly toxic gases may be emitted with combustion or thermal decomposition. Such a threat is aggravated in its finely divided state as it may be ignited by sparks alone. Few potential health hazards are –Ingestion can be particularly harmful causing irritation in digestive tractInhalation can cause irritation in respiratory tract as well as mucous membranesMoreover, owing to the flammable nature, dimethylglyoxime is a direct fire hazard. Which of the following statement is incorrect?(a) There is tetrahedral geometry in the red complex(b) There is symmetrical hydrogen bonding in the complex(c) There is square planar geometry in the red complex(d) Dimethylglyoxime acts as a bidentate ligandDMG can cause severe health effects in case of unregulated exposure. Xbox 360 black ops 2 save editor for macHence, when you have a clear understanding of such topics, you can hit the ground running in terms of preparation. Join our online classes to revisit the basics of the subject as well as for greater discussion on important areas such as dimethylglyoxime and its properties.Questions from certain topics, such as DMG structure, are frequently asked in examination. It is better to identify the areas of weakness in the early stage of your preparation.
0 Comments
Leave a Reply. |
AuthorAnthony ArchivesCategories |
 RSS Feed
RSS Feed
